Models
In the manual science teachers can find practical steps and examples of how to use the developed pedagogical tools - models, examples and experiments in their everyday educational process. The manual is based on the approach for development of comprehension in science education, in particular the development of reverse thinking and the fact that its existence is a direct sign of understanding. The manual aims to significantly ease the the job of teachers and give in their hands ready-to-use tools to support learning of students, since it is quite a difficult job to create the right models for each science concept and it may take too long to explain without them the new study subject.
A covalent bond
 Scientific definition
Scientific definitionA covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs ...
Acid / base indicator
Acid - Base indicators (also known as pH indicators) are substances which change colour with pH. They are usually weak acids or bases, which when ...
Acid / base indicator
 Scientific definition
Scientific definitionAcid - base indicators (also known as pH indicators) are substances which change color with pH. They are usually weak acids or bases. ...
Acid-base indicators
 Scientific definition
Scientific definitionThe acid-base indicators, or pH indicators, are organic substances that have the property of assuming different colors below or above a determined ...
Amino acids
 Scientific definition
Scientific definitionAmino acid - any of a large number of compounds found in living cells that contain carbon, oxygen, hydrogen, and nitrogen, and join together to fo ...
Amount of substance
 Scientific definition
Scientific definitionIn every amount of a substance, there is a enormously big number on particles, ions or molecules. In order to simplify calculation with them ...
An electron shell
 Scientific definition
Scientific definitionAn electron shell is a grouping of electrons surrounding the nucleus of an atom. ...
An electron shell (2)
 Scientific definition
Scientific definitionAn electron shell is a grouping of electrons surrounding the nucleus of an atom. ...
Anions
 Scientific definition
Scientific definitionAnions are atoms or radicals (groups of atoms), that have gained electrons. Since they now have more electrons than protons, anions have a negativ ...
Catalyst
 Scientific definition
Scientific definitionA catalyst is a substance that enables a chemical reaction to proceed at a usually faster rate or under different conditions (as at a lower temper ...
Catalyst (2)
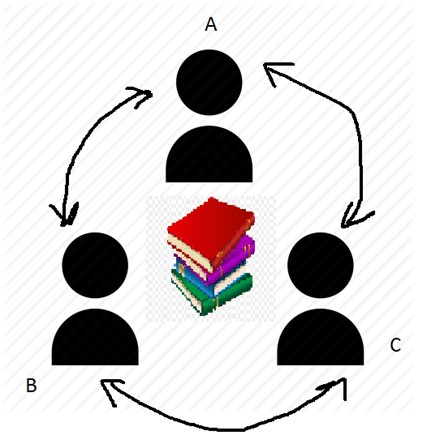 Scientific definition
Scientific definitionA catalyst is a substance that enables a chemical reaction to proceed at a usually faster rate or under different conditions (as at a lower temper ...
Cations
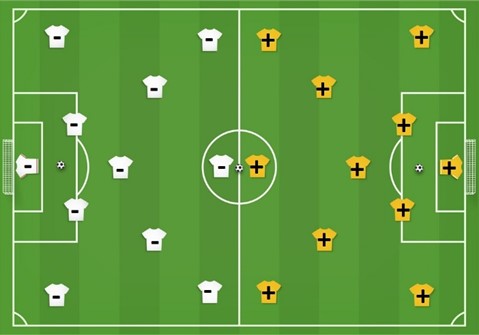 Scientific definition
Scientific definitionCations are atoms or radicals (groups of atoms), that have lost electrons. Since they now have more protons than electrons, cations have a positiv ...
Chemical bond
The chemical bond corresponds to the force of attraction that holds together the atoms that are part of a molecule, to achieve stability. ...
Chemical equilibrium
 Scientific definition
Scientific definitionIn a chemical reaction, chemical equilibrium is the state in which both reactants and products are present in concentrations, which have no fur ...
Chemical reactions. Signs of chemical reactions
Chemical reactions are the processes by which chemicals interact to form new chemicals with different compositions. Simply stated, a chemical reac ...
Dalton’s Law
Dalton's law says that when one element combines with another to form different compounds, the masses of the elements stand between them in re ...
There are no links
Diffusion
 Scientific definition
Scientific definitionDiffusion is the movement of a substance from an area of high concentration to an area of low concentration. Diffusion happens in liquids and ...
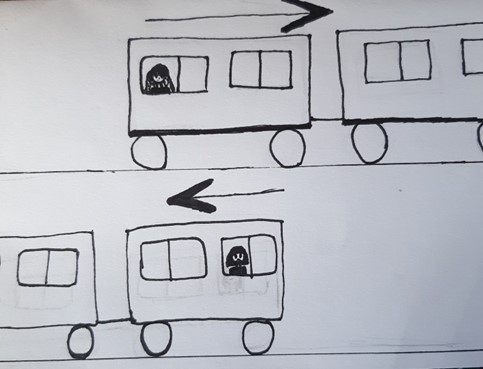
Diffusion
 Scientific definition
Scientific definitionDiffusion is the movement of a substance from an area of high concentration to an area of low concentration. Diffusion happens in liquids and ...
Distillation
Distillation is the physical process of separating the components or substances from a liquid mixture by using selective boiling and condensation. ...
DNA composition
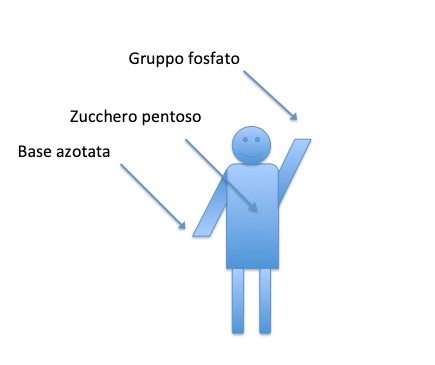 Scientific definition
Scientific definitionThe DNA molecule consists of two chains of nucletids. Each nucleotide consists of a pentose sugar, a phosphate group, a nitrogenous base. The nuc ...
There are no links
Effective collision (The collision theory)
A collision between two reactants ( molecules, atoms or ions = In general terms: particles of r.) with the correct orientation and with sufficient ...
Electrolysis
Electrolysis is the chemical process of using an electrical current to stimulate non-spontaneous reactions. A non-spontaneous reaction is one t ...
Electrolytes and conduct electricity electrolytes
Electrolytes are composed of free moving Charged Particles, i.e. ions. When a pd is applied across the electrolytes, the positive ions move toward ...
Electrolytes, Battery
An electrolyte is a substance that produces an electrically conducting solution when dissolved in a polar solvent, such as water. The dissolved el ...
Enzymes
 Scientific definition
Scientific definitionEnzymes are macromolecules that helps speed up chemical reactions in the organism‘s body. They break down molecules such like proteins, carbohy ...
There are no links
Esterification
 Scientific definition
Scientific definitionEsterification is chemical reaction that forms at least one ester (= a type of compound produced by reaction between acids and alcohols). Esters a ...
Hydrolysis
 Scientific definition
Scientific definitionHydrolysis, the opposite of condensation, is a chemical reaction in which water breaks down another compound and changes its makeup. Most instances ...
Indicator
 Scientific definition
Scientific definitionChemical indicator - any substance that gives a visible sign, usually by a colour change, of the presence or absence of a threshold concentration ...
Ionic bond
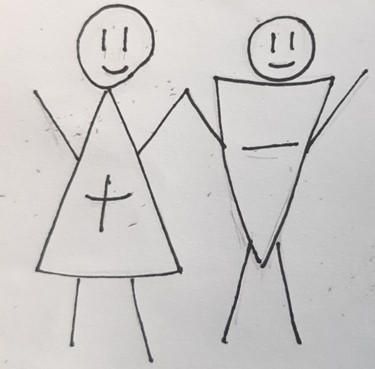 Scientific definition
Scientific definitionThe definition of ionic bond is when a positively charged ion forms a bond with a negatively charged ions and one atom transfers electrons ...
Iron
Iron is one of the most common metal elements that is extracted from minerals to produce cast iron and subsequently steel.
There are no links
Isomery
 Scientific definition
Scientific definitionIn chemistry, isomers are ions or molecules with identical formulas but distinct structures. Isomers do not necessarily share similar propert ...
Isotopes
 Scientific definition
Scientific definitionIsotopes are atoms that have the same number of protons and electrons but different numbers of neutrons and therefore have different physical prop ...
Isotopes (2)
 Scientific definition
Scientific definitionIsotopes are atoms that have the same number of protons and electrons but different numbers of neutrons and therefore have different physical prop ...
Isotopes (3)
 Scientific definition
Scientific definitionAn isotope is the form of an atom which has a different atomic weight from other forms of the same atom, but the same chemical structure (two or m ...
Molecules
Solid matter is composed of tightly packed particles. Liquid matter is made of more loosely packed particles. Particles can move about within a li ...
There are no links
Osmosis
Osmosis is the spontaneous net movement of solvent molecules through a selectively permeable membrane into a region of higher solute concentration ...
Periodic system
 Scientific definition
Scientific definitionThe periodic system is a tabular disposition of the chemical elements, ordered by their atomic number, electronic configurations and recurrent che ...
Polymerization process
Polymerization: Polymerization of olkenes: polyaddition chain reaction. A collision between two reactants ( molecules, atoms or ions = In ...
Proton Neutron Electron
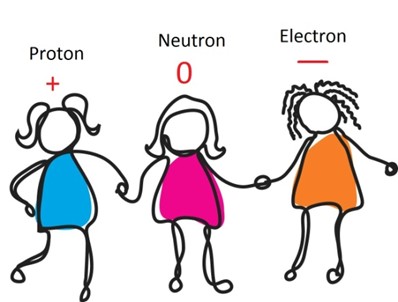 Scientific definition
Scientific definitionA proton is an atomic particle that has a positive electrical charge. A neutron is very small piece of matter, i.e. substance, that carries no ...
There are no links
Redox reactions
Traffic light is the name of a clock reaction which will spontaneously change color from yellow to red after a gentle shake. After a more vigoro ...
Sublimation
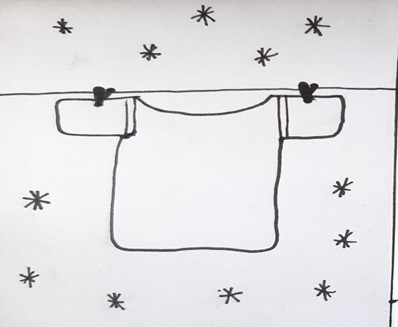 Scientific definition
Scientific definitionSublimation is the transition of a substance directly from the solid to the gas phase, without passing through the intermediate liquid phase. ...
Substitution reaction
 Scientific definition
Scientific definitionSubstitution reaction (also known as single displacement reaction or single substitution reaction) is a chemical reaction during which one functio ...









